Transcription-dependent metabolic plasticity in neurons
We and others recently discovered a synaptic activity- and calcium-regulated gene program that drives changes in neuronal energy metabolism (Bas-Orth et al., JBiolChem 2017; Segarra-Mondejar et al., EMBOJ 2018). In particular, we found that glucose metabolism shifts from oxidative phosphorylation towards aerobic glycolysis in synaptically activated neurons. This novel form of metabolic plasticity, which we termed ‘neuronal Warburg effect’, might be important for mitochondrial homeostasis and neuroprotection. We are now using metabolomics approaches to obtain a more comprehensive understanding of activity-dependent metabolic reprogramming beyond glucose metabolism. In addition, we are using live imaging of fluorescent nanosensors for glucose, ATP, and redox potential to study subcellular adaptations of neuronal energy metabolism.
We and others recently discovered a synaptic activity- and calcium-regulated gene program that drives changes in neuronal energy metabolism (Bas-Orth et al., JBiolChem 2017; Segarra-Mondejar et al., EMBOJ 2018). In particular, we found that glucose metabolism shifts from oxidative phosphorylation towards aerobic glycolysis in synaptically activated neurons. This novel form of metabolic plasticity, which we termed ‘neuronal Warburg effect’, might be important for mitochondrial homeostasis and neuroprotection. We are now using metabolomics approaches to obtain a more comprehensive understanding of activity-dependent metabolic reprogramming beyond glucose metabolism. In addition, we are using live imaging of fluorescent nanosensors for glucose, ATP, and redox potential to study subcellular adaptations of neuronal energy metabolism.
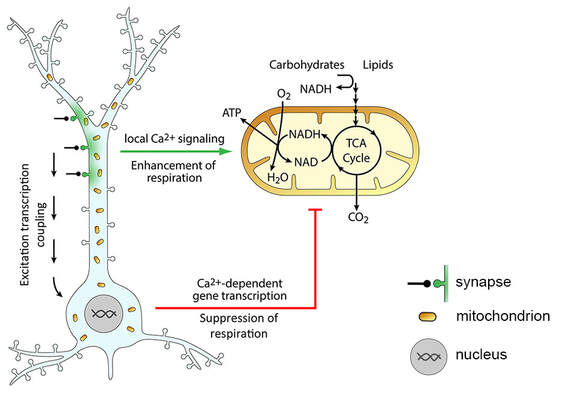
Figure 1. Activity-dependent metabolic plasticity in neurons. Synaptic activity leads to influx of Ca2+ (green) into the postsynaptic compartment, where it locally activates mitochondrial respiration to enhance ATP production. Strong or sustained synaptic activity triggers changes in gene transcription (excitation transcription coupling) that lead to a decrease of mitochondrial respiration, most likely to preserve mitochondrial homeostasis. Adapted from Bas-Orth et al., 2017.
|
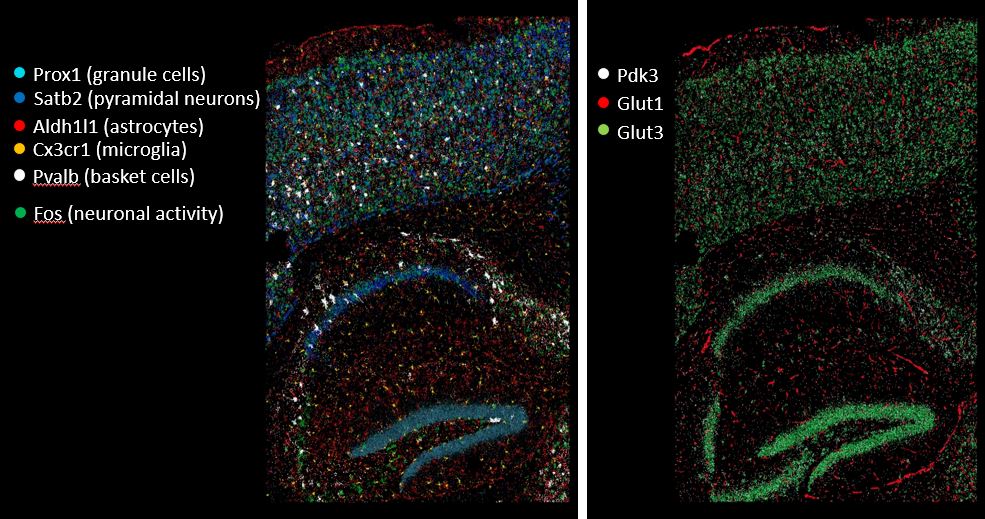
Figure 2. Spatial gene expression analysis. We use combinatorial single-molecule fluorescent in-situ hybridization (smFISH) in sections of the mouse hippocampus to study cell type- and region-specific regulation of metabolic gene expression upon neuronal activity in vivo. 8 out of a total of 35 analyzed transcripts are illustrated.
|
Mitochondria as targets for neuroprotective strategies in neuroinflammation
Mitochondria are integral for cellular function and survival. They generate ATP for cellular energy homeostasis, act as a Ca2+ sink and source to influence cellular Ca2+ signaling, and are key hubs for necrotic and apoptotic signaling networks. Accordingly, aberrant mitochondrial function is key to many neurodegenerative and neuroinflammatory diseases. In this project, we investigate the relationship between mitochondrial Ca2+ signaling, mitochondrial structure and function, and synaptic integrity in a model of peripheral inflammation. We are using immunofluorescence and enzyme histochemistry to analyze mitochondrial structure and function in the brains of healthy and inflamed mice. By manipulating the expression levels of mitochondrial Ca2+ transporter proteins we aim at restoring mitochondrial Ca2+ homeostasis, metabolic function, and synaptic activity in this disease model.
Mitochondria are integral for cellular function and survival. They generate ATP for cellular energy homeostasis, act as a Ca2+ sink and source to influence cellular Ca2+ signaling, and are key hubs for necrotic and apoptotic signaling networks. Accordingly, aberrant mitochondrial function is key to many neurodegenerative and neuroinflammatory diseases. In this project, we investigate the relationship between mitochondrial Ca2+ signaling, mitochondrial structure and function, and synaptic integrity in a model of peripheral inflammation. We are using immunofluorescence and enzyme histochemistry to analyze mitochondrial structure and function in the brains of healthy and inflamed mice. By manipulating the expression levels of mitochondrial Ca2+ transporter proteins we aim at restoring mitochondrial Ca2+ homeostasis, metabolic function, and synaptic activity in this disease model.
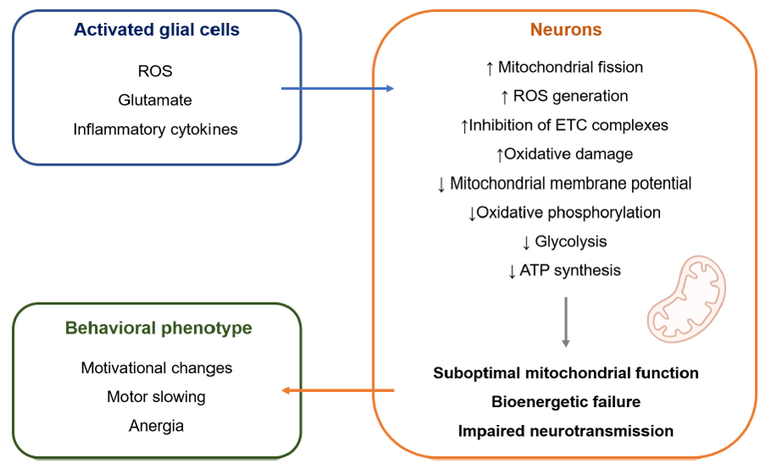
Figure 3. Proposed role of mitochondrial dysfunction in inflamed depression. Peripheral inflammation in the body can spread to the brain via cellular, humoral and neural routes, leading to activation of glial cells. Activated glial cells release soluble mediators that affect mitochondrial function in nearby neurons. This might result in bioenergetic impairment of neurons, leading to disturbed synaptic function as an underlying cause of inflammation-induced depression. Figure is from Casaril, Dantzer, Bas-Orth (2021) Front Neurosci 15:725547
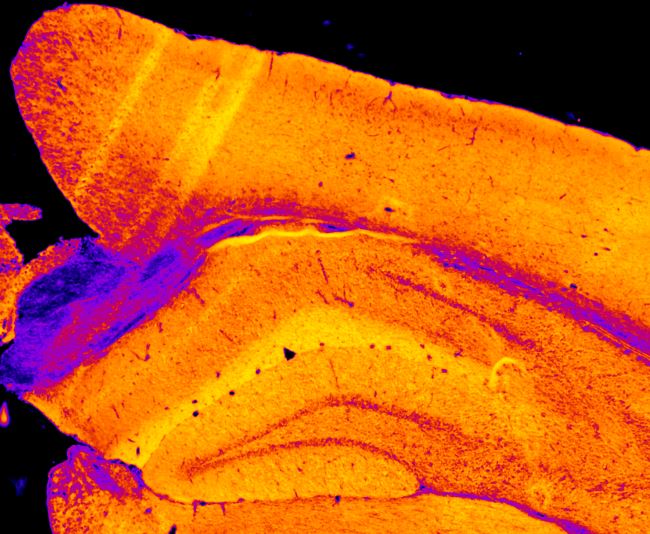
Figure 4. Enzyme histochemistry to assess mitochondrial function in the brain. Activity of cytochrome c oxidase (mitochondrial respiratory complex IV) was measured in a mouse brain section. Color scale indicates enzyme activity (arbitrary units).